Screening for good ligand candidates requires multiple steps, one of which is to evaluate their binding properties. The use of fluorescence spectroscopy can provide important information about the process of ligand binding and ligand-dependent conformational changes in receptors, particularly the kinetic aspects of these processes, which are difficult to extract from x-ray structures. Fluorescence-based binding assay is a promising alternative to traditional radioactive assay. Firstly, the fluorescent probes needed for such tests are safe to use, and they do not have the same limitations as radioactive probes, making them easier to implement. Secondly, bright and stable probes have been developed and are now commercially available. Thirdly, potent fluorescent ligands have been designed and synthesized for numerous receptors due to the structure/activity relationship analysis. Finally, instruments needed to measure fluorescence are also becoming more and more sensitive and are frequently used to perform common fluorescence or luminescence-based assays.
Fluorescence has the advantage that only a small amount of target protein and ligand are required, which can be easily automated and even low affinity binding can be characterized. Fluorescence has a broad spectrum of wavelengths, therefore multiple colors can be applied to detect a specific target. At present, there are many methods for measuring fluorescence intensity. Less hazardous, faster and cheaper, these methods also exhibit very good sensitivity and can be used on various biological models such as heterologous expression systems or native tissues. In addition to changes in fluorescence intensity, changes in emission peak position and steady-state anisotropy may also vary with the binding of a ligand, which can be used to obtain binding constants.
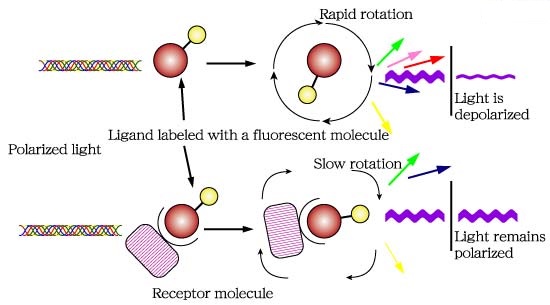 Figure 1. Diagram of a fluorescence polarization assay.
Figure 1. Diagram of a fluorescence polarization assay.
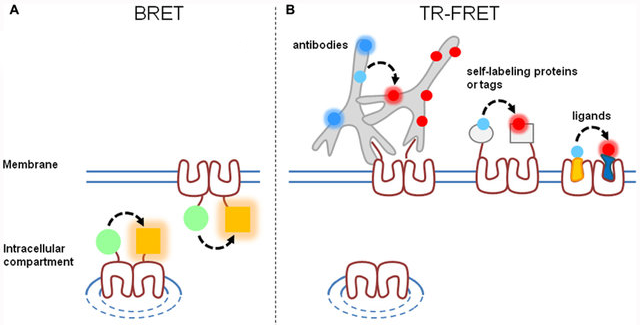 Figure 2. Comparison of BRET and time-resolved FRET approaches.
Figure 2. Comparison of BRET and time-resolved FRET approaches.
- Fluorescence polarization (FP)
- Fluorescence and bioluminescence resonance energy transfer (FRET and BRER)
- Fluorometric microvolume assay technology (FMAT)
- Fluorescence correlation spectroscopy (FCS)
- Flow cytometry
At Creative Bioarray, our fluorescent ligand binding assays offer the ability to:
- Detect and quantify target proteins localized to the cell surface
- Perform and detect cell surface biotinylation
- Monitor receptor internalization and recycling
- Evaluate the effects of mutations, drugs, and other treatments on protein trafficking
- Quantify ligand binding to cell surface receptors
- Analyze multiple samples quickly and quantitatively
- Avoid use of radioactive materials
As an experienced drug development company, we are committed to using our expertise and innovative technology platform to effectively save you the cost and time of GPCR binding studies. The bioscientists at Creative Bioarray have extensive experience in developing a wide range of binding assays in a variety of formats to ensure your project has a tailored solution. If you have any needs, please feel free to contact us.
References
- Cottet M. et al.; BRET and Time-resolved FRET strategy to study GPCR oligomerization: from cell lines toward native tissues. Frontiers in Endocrinology, 2012, 3: 92.
- De Jong L. A. A. et al.; Receptor-ligand binding assays: Technologies and Applications. Journal of Chromatography B, 2005, 829: 1-25.