It is well known that GPCR activation can alter gene transcription through responsive elements for second messengers, such as cAMP response element (CRE), nuclear factor of activated T-cells response element (NFAT-RE), serum response element (SRE), and serum response factor response element (SRF-RE, a mutant form of SRE), all of which are located in the promoter region of the gene. Therefore, cell-based reporter assays provide another popular and cost-effective HTS platform for GPCR screening. The reporter gene assays have the advantages of wide linearity, high sensitivity and large signal-to-background ratio, and is suitable for detection of weak GPCR agonists or allosteric modulators. Reporter gene assays are also easy to set up and can be scaled down to extremely low assay volumes in 1536- or 3456-well formats.
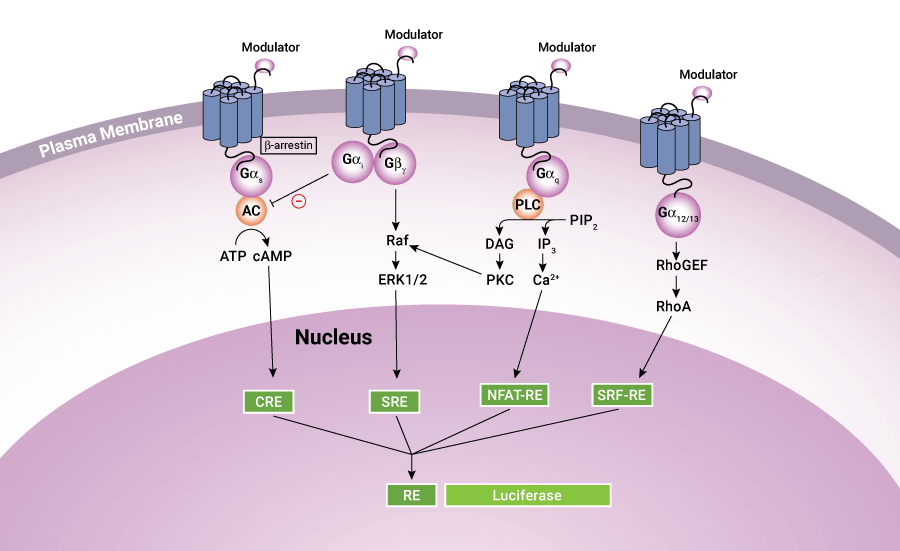 Figure 1. Schematic diagram showing GPCR signaling pathways.
Figure 1. Schematic diagram showing GPCR signaling pathways.
Reporter Assays at Creative Bioarray
In our reporter assays, GPCR signal transduction is monitored using an expression system engineered with cis-acting enhancer elements, DNA sequence motifs targeted by binding partners and upstream of the reporter gene. Commonly used reporters are enzyme proteins with activities linked to a variety of colorimetric, fluorescent or luminescent readouts, such as luciferase, alkaline phosphatase, β-galactosidase, β-lactamase and various fluorescent proteins. Among them, luciferase has become the preferred reporter for high-throughput screening due to its high sensitivity, broad dynamic range, lack of endogenous activity and low interference coming from the compounds. In addition, specially developed detection reagents allow convenient measurement of two luciferase activities in the same sample.
| Reporter | Detection Method | ||
| Luminescence | Fluorescence | Absorbance | |
| Luciferase | + | + | |
| β-Galactosidase (β-Gal) | + | + | + |
| β-Glucuronidase (β-GUS) | + | + | |
| Secreted alkaline phosphatase (SEAP) | + | + | |
| Green Fluorescent Protein (GFP) | + | ||
Applications of Reporter Assay
- Characterize the strength of promoters and enhancers
- Help in defining or characterizing role of various transcription factors
- Measures transfection efficiency and the success of molecular cloning
- Monitoring expression of gene and protein trafficking
References
- Zhang R. et al.; Tools for GPCR drug discovery. Acta Pharmacologica Sinica, 2012, 33(3): 372-384.
- Cheng Z. et al.; Luciferase reporter assay system for deciphering GPCR pathways. Curr. Chem. Genomics, 2010, 4: 84-91.
- Receptor Binding Assay
- GTPγS Binding Assay
- cAMP Assay
- Ca2+ Mobilization Assay
- IP3/IP1 Assay
- GPCR Internalization Assay
- Label-free Whole Cell Assay
- Receptor Dimerization Assay