At present, the advantages of GPCRs as molecular targets have gradually weakened in newly developed drugs, in part because routine screening tests have been exhausted. Therefore, new screening assays, such as heterodimer-targeted ligand screening, are needed for further drug discovery. It is worth noting that dimerization can increase the diversity of GPCR signaling regulation, and thus the specific evaluation of signaling characteristics in different dimerized receptors is of great significance, not only for the development of new drugs, but also for understanding signal networks.
Evidence for GPCR dimerization comes from biochemical, biophysical and functional studies. Two receptors form a dimer through the interaction of their extracellular loops, transmembrane helices and intracellular loops. The nature of bonds between two receptors can vary from covalent to non-covalent or a combination of both.
Dimerization can occur in and affect different stages of the receptor’s life, namely trafficking, signaling and internalization, and can be seen as a natural way to regulate receptor activity or increase the library of protein functions. Therefore, compounds directed against GPCR heterodimers or compounds that affect receptor dimerization may have the potential to achieve specific therapeutic effects. As a result, it is meaningful to design assays to assess the effect of compounds on GPCR dimerization.
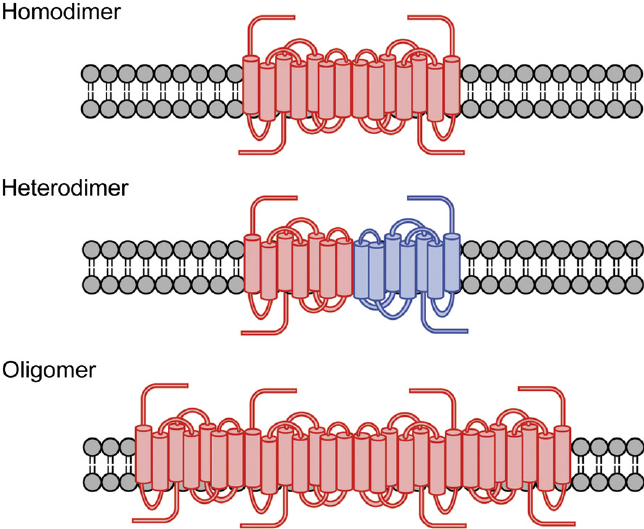 Figure 1. Formation of GPCR dimers and oligomers.
Figure 1. Formation of GPCR dimers and oligomers.
Receptor Dimerization Assays at Creative Bioarray
Creative Bioarray has established a variety of technologies to monitor receptor dimerization, including those based upon traditional biochemistry such as blue-native polyacrylamide gel-electrophoresis (BN-PAGE), co-immunoprecipitation and protein-fragment complementation assays, those based upon resonance energy transfer, fluorescence resonance energy transfer (FRET), time-resolved FRET, FRET spectrometry and bioluminescence resonance energy transfer (BRET), as well as those based upon microscopy such as fluorescence recovery after photo-bleaching (FRAP), total internal reflection fluorescence microscopy (TIRF), spatial intensity distribution analysis (SpIDA) and various single molecule imaging techniques.
References
- Guo, H. et al.; Methods used to study the oligomeric structure of G-protein-coupled receptors. Bioscience Reports, 2017, 37(2): BSR20160547.
- Lin, H. et al.; Cofactoring and dimerization of proteinase-activated receptors. Pharmacological Reviews, 2013, 65(4): 1198-1213.
- Receptor Binding Assay
- GTPγS Binding Assay
- Reporter Assay
- cAMP Assay
- Ca2+ Mobilization Assay
- IP3/IP1 Assay
- GPCR Internalization Assay
- Label-free Whole Cell Assay