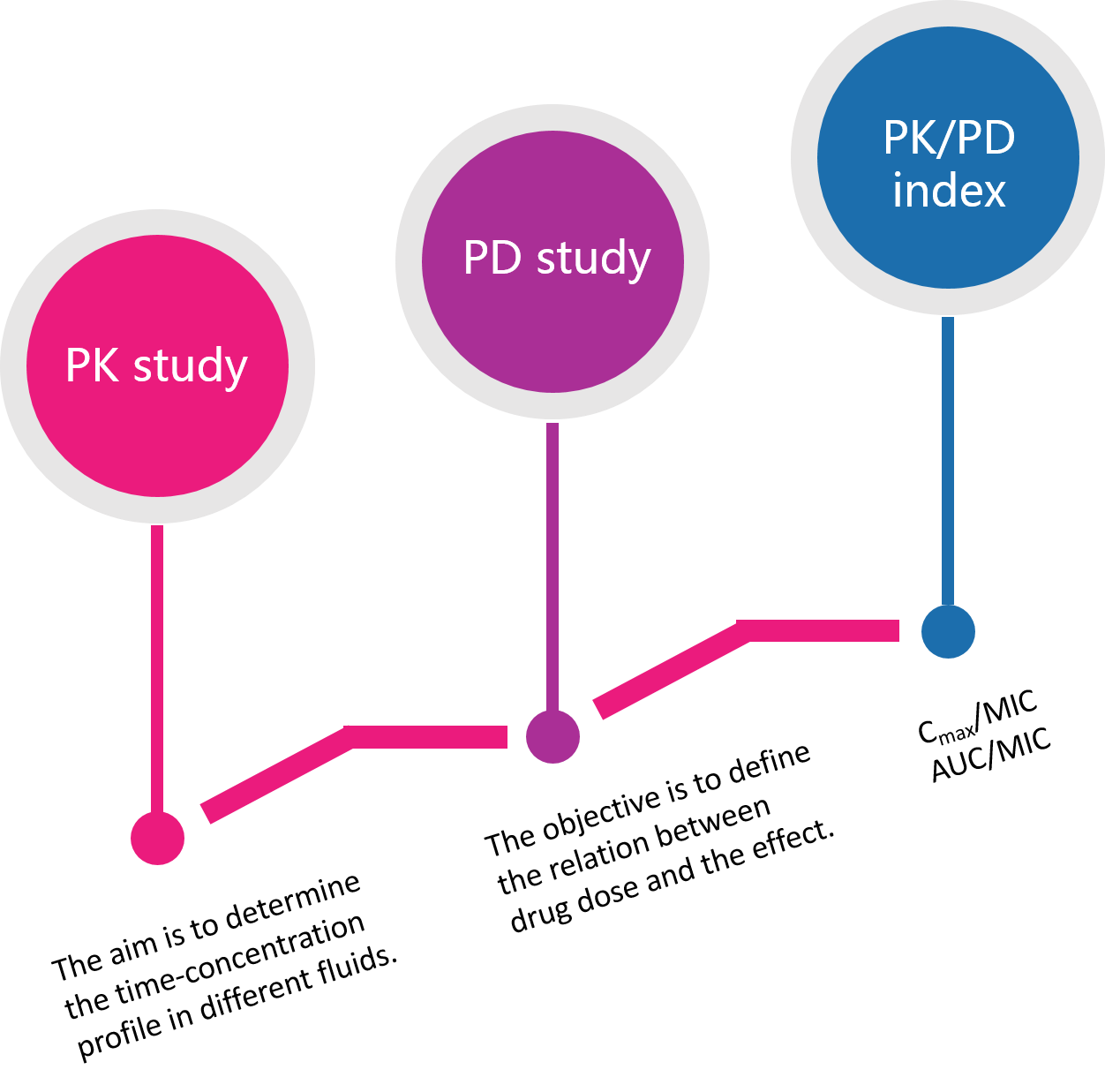
Pharmacodynamics (PD) refers to the action of a drug to the body, involving receptor binding, post-receptor effects, and chemical interactions. Pharmacokinetics (PK) determines the onset, duration, and intensity of drug action. Successful drug discovery relies on the selection of drug candidates with good in vivo PK properties, as well as appropriate preclinical efficacy and safety. In vivo PK profiling is often a bottleneck in the discovery process.
In order to prove effective, a drug delivered to the patient must reach the necessary concentration in the plasma and in relevant tissues (as measured by PK). This is necessary to effectively modulate the activity of the target protein (as measured by PD) in the body. Incorporating in vivo PK/PD efficacy studies at the early stages of drug development program can significantly accelerate the selection of the most promising compounds. The rationales for conducting in vivo PK/PD efficacy studies in preclinical models are to:
- provide a quantitative analysis of the dose-response relationships;
- describe and predict the time-course effects of one or multiple drug doses;
- understand the frequency and the extent of target modulation to observe statistically significant efficacy;
- determine efficacious dosing regimens with an acceptable therapeutic index.
PK/PD analysis is typically performed at every stage of the drug development process. As development is becoming more complex, time consuming, and cost intensive, Creative bioarray is looking to make better use of PK/PD data to eliminate defective candidates from the outset and identify those most likely to achieve clinical success.
Our services for in vivo assessment of PK/PD properties include but are not limited to:
- Standard formulation and specific formulation development
- Formulation screening
- Allometric dose prediction and PK optimization
- Single, multiple, and cassette dosing PK studies
- BBB penetration (rat/mouse)
- Whole blood-to-plasma partitioning
- Stability in blood, plasma, gastric and intestinal fluids
- Mass balance and tissue distribution (traditional method and QWBA)
- Entero-hepatic recirculation (EHC)
- Renal and biliary excretion
Available species
Mouse, Rat, Hamster, Guinea pig, Rabbit, Beagle dog, Mini-pig and Cynomolgus monkey.
Administration routes
Oral (PO), intravenous (IV), intraarterial (IA), intramuscular (IM), intraperitoneal (IP), intratracheal (IT), subcutaneous (SC), transdermal (TD), intranasal (IN), IV infusion, SC infusion, rectal, and topical administration.
Surgical techniques
Bile-duct cannulation (BDC), jugular cannulation (JVC), portal vein cannulation (PVC), carotid artery cannulation (CAC), mesenteric lymph duct cannulation, bile-duct ligation, and castration.
Sample types
Whole blood, plasma, serum, bile, feces, urine, cerebrospinal fluid, bone marrow, lymph node, other fluids and tissues.
The features of our PK/PD services
- AAALAC-accredited animal facilities for a wide range of animal species
- State-of-the-art instruments
- Extensive experiences in PK model development with comprehensive in vivo techniques (surgical operation, animal care)
- Formulation support including formulation screening, development as well as formulation recommendation for various species and strains
- PK/PD data analysis, modeling, and simulation
- Regulatory documents support
PK/PD analysis is a key aspect of drug development. Understanding the bioavailability, exposure, half-life, clearance, and metabolism of a drug may determine the success or failure in the clinic. Creative Bioarray offers in vivo PK/PD services by utilizing our extensive expertise and models to support drug discovery and preclinical development for small molecules, peptides, nucleosides/nucleotides and biologics. All in-life work of PK/PD studies are carried out in AAALAC-approved animal facilities and follow IACUC-approved protocols. Whatever your needs, we work with you to identify the most appropriate models and to provide the customized study designs.
- In Vitro ADME
- Metabolite Profiling and Identification
- Bioanalytical Services
- BA/BE Services
- Quantitative Tissue Distribution